BIP – Laboratoire de Bioénergétique et Ingénierie des Protéines

The labs
- AFMB - Architecture et Fonction des Macromolécules Biologiques
- BBF - Biodiversité et Biotechnologie Fongiques
- BIAM - Bioscience and Biotechnology Institute of Aix-Marseille
- BIP - Laboratoire de Bioénergétique et Ingénierie des Protéines
- IGS - Laboratoire Information Génomique et Structurale
- IMM - Institut de Microbiologie de la Méditerranée
- iSm2 - Institut des Sciences Moléculaires de Marseille - BiosCiences
- LCB - Laboratoire de Chimie bactérienne
- LISM - Laboratoire d'Ingénierie des Systèmes Macromoléculaires
- MCT - Membranes et Cibles Thérapeutiques
- MIO - Mediterranean Institute of Oceanography - Environmental Microbiology and Biotechnology
Key Figures
- 7 Teams
- 21 Researchers
- 13 Teacher-researchers
- 8 Engineers/Technicians
- 18 Doctoral students
- 11 CDD
Research
The Bioenergetics and Protein Engineering lab (BIP – UMR 7281) is a Joint Research Unit of the CNRS, under agreement with Aix-Marseille University (AMU). BIP is the founding member of the Institut de Microbiologie de la Mediterranée and of the IM2B, BIP is a member of the Federation of Chemistry of Marseille.
BIP’s research themes focus on exploring the diversity of energy metabolisms in microorganisms and their applications in the field of bioenergy and the environment. Bringing together biologists, chemists and physicists, the BIP is developing an original approach based on the latest advances in structural genomics, functional proteomics and molecular biophysics. This remarkable multidisciplinary enables an integrated approach to energy conversion processes, from the physiological level to the molecular bases responsible for the enormous variability of substrates and reactivity in the enzymes and supramolecular structures involved. It also makes it possible to trace the evolution of these systems from the earliest bioenergetic mechanisms to their current diversity.
Highlighting metabolic biodiversity, understanding the adaptation and resistance of microorganisms to extreme conditions, and deciphering fundamental metabolisms are important issues. Their understanding at the fundamental level is also at the source of biotechnological innovations essential for the search for new energy sources and for sustainable development: biogas and hydrogen production, biomass, bioremediation, bioconversions, bacteria-environment interactions.
A benchmark laboratory through its achievements in environmental microbiology, BIP is one of the strong players in the « Capénergies » competitiveness cluster and has established fruitful industrial partnerships (HTS-Bio, Veolia, ENGIE….).
The laboratory is organised into 7 research teams each with their specific thematic.
The Teams
Adaptation systems of bacteria – Heads: Marie-Thérèse Giudici-Orticoni
How do bacteria adapt to changing environments? From the molecular to the cellular scale.
The fascinating ability of bacteria to thrive in changing environments lies in complex adaptation systems. In our group, we aim at understanding the mechanisms by which bacteria (i) modify their metabolism to use available resources, (ii) reorganize their genetic expression using alternative transcription factors, (iii) ensure the proper folding and protection of proteins in function of stress, (iv) move towards nutrients or flee toxic compounds, and at last (v) adapt at the population level by forming biofilms or by using resources produced by other species within bacterial consortia. These studies are conducted at different scales, from the molecular to the cellular scale, and even to the multi-cellular scale.
Enzymology of supramolecular systems – Heads: Brigitte Gontero
From CO2 acquisition in algae to lipid metabolism
We are a group of enzymologists, biologists, bio-chemists, bio-physicists, structural biologists working on micro-algal metabolism: from CO2 uptake to lipid remobilization. We study this using a range of experimental and computational methods on different strains of microalgae. We have a special interest in flexible proteins involved in CO2 metabolism, in CO2 concentrating mechanisms that optimize carbon fixation in aquatic environments and enzymes involved in the hydrolysis of galactolipids which are present in chloroplast membranes. We welcome students from different levels and fields and have a vibrant lab with visiting scientists from abroad.
Hydrogen metabolism – Heads: Myriam Brugna
Understanding microbial H2 metabolism and hydrogenases
Our research focuses on hydrogenases, which are enzymes that reversibly catalyze the oxidation of molecular hydrogen into protons. The goal of our work is to study the role of hydrogenases in the energy metabolism of microorganisms as well as the reaction mechanisms of these enzymes. We are thus developing an integrated multidisciplinary approach to hydrogen metabolism, in close collaboration with groups of chemists and physicists of the laboratory, from the enzyme and the catalytic activity to its cellular functions and its role in energy-generating mechanisms. Our work is therefore part of the laboratory’s « hydrogen and associated metabolisms » thematic axis. Our research also aims to remove the scientific barriers to the use of hydrogenases in biotechnological processes and in particular to understand the molecular basis of the inactivation of hydrogenases by O2, which is one of the main current limitations.
Reaction dynamics of multicenter redox enzymes, electrochemical kinetics – Heads: Christophe Léger
Studying structure-function relationships in biological redox catalysis
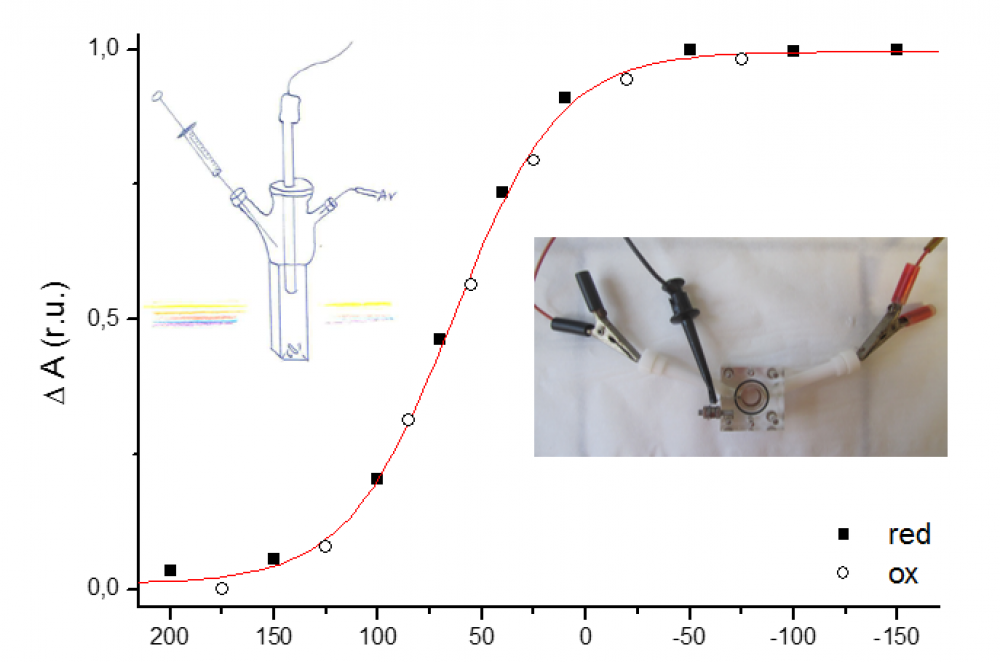
We investigate the mechanism of multicenter redox enzymes by combining various kinetic techniques, including direct electrochemistry. By “direct electrochemistry”, we mean that the enzyme is adsorbed onto a rotating electrode which is immersed into a solution of the substrate, electron transfer between the enzyme and the electrode is direct, and the activity is simply monitored as a current. The data can be used to gain information about virtually every step in the mechanism. We work on various redox enzymes, particularly molybdoenzymes and hydrogenases, in collaboration with several biochemistry groups. We are interested in studying long distance electron transfer in multicenter redox enzymes, the mechanism at the active site of these enzymes, the oxygen sensibility of hydrogenases. We seek to understand the molecular basis of some of the global properties of redox enzymes: this includes catalytic bias, substrate specificity and resistance to chemical stress.
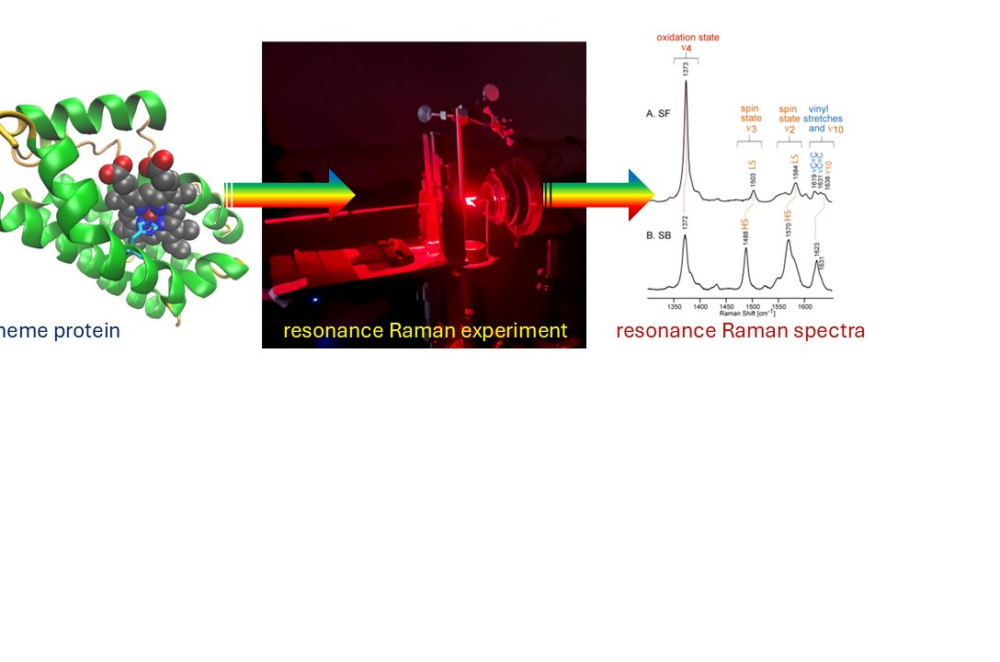
Biophysics of metalloproteins – Heads: Bruno Guigliarelli
Sujet d’équipe
Bioelectrochimistry, biointerfaces and biotechnologies – Heads: Elisabeth Lojou
Mimicking electron physiological transfers to ensure bioelectrocatalysis
Our main objective is to get inspiration from bacterial electron transfer pathways to ensure efficient bioelectrocatalysis. We consider both planar and nanostructured electrodes (deposited nanoparticles in particular) that we chemically modified to favor a functional protein binding. By coupling electrochemisty to molecular modeling, spectroscopies, and microscopies we aim to get a full understanding of the relationship between protein loading, conformation and electroactivity. These fundamental studies at the interface between biochemistry and physico-chemistry open applicative perspectives in biosensing or bioenergy generation.
Evolution of bioenergetics – Heads: Barbara Schoepp-Cothenet
What are the origins of biological energy conversion systems and how did they evolve from the Last Unique Common Ancestor to now?
All forms of Life rely on conversion of redox energy into proton motive force (pmf) and ATP. In our group, we study selected prokaryotic bioenergetic chains that lead to the build-up of this pmf, with the aim of contributing to a comprehensive picture of the diversity of these chains but also of elaborating common features and conserved principles. To this end, our team brings together expertise from phylogeny, biochemistry, molecular biology and biophysics for studies of the reactions occurring at the active site of the enzymes, on a molecular level. Because the ultimate goal of our work is to contribute to the elucidation of the origin(s) and evolutionary pathways of biological energy conversion, we furthermore address the origin of life from minerals and the imprint this origin left in current redox enzymes. To this end, we collaborate with geochemists and physicists for studies of clay minerals’ catalysis.
Proposed Trainings
BIP contacts
BIP website- bip-sec@imm.cnrs.fr
- +33 4 91 16 45 05
-
BIP (UMR 7281)
31 Chemin Joseph Aiguier – CS70071
13402 Marseille Cedex 09 – FRANCE